Welcome from EUS-AAEM
The Emergency Ultrasound Section of the American Academy of Emergency Medicine (EUS-AAEM) was founded to foster the professional development of its members and to educate them regarding point-of-care ultrasound. This group will serve as a venue for collaboration among medical students, residents and practitioners who are interested in point-of-care ultrasound. The purpose of our group is to augment the knowledge and expertise of all emergency medicine specialists and to advocate for patient safety and quality care by endorsing bedside ultrasound. Membership is not limited to fellowship trained physicians. All emergency medicine practitioners passionate about ultrasound are welcome to join and participate.
We are proud to publish our e-newsletter with original contributions from many of our members. We encourage all members to submit for future editions. Topics include but are not limited to educational, community focus, interesting cases, resident and student section, and adventures abroad.
For more information, visit our webpage.
In this Issue
Original POCUS Research
SUPER Protocol: A Structured POCUS Approach to Source Identification in Undifferentiated Sepsis
By Sreekara, VR MBBS and Amrithanand, VT MD
Introduction
Sepsis is a time-critical emergency with high morbidity and mortality and early identification of the infection source is fundamental to effective management and improved patient care outcomes¹. In the emergency department (ED), many patients present with undifferentiated sepsis and the history and physical examination may fail to localize the source. Diagnostic delays often result in delayed initiation of appropriate treatment, prolonged empiric broad-spectrum antibiotic use that can lead to antibiotic resistance, and lack of infectious source control³.
Point-of-care ultrasound (POCUS) evaluates internal organs and structures at the bedside, and studies have shown expedited diagnosis and improved accuracy of sepsis source identification compared with clinical evaluation alone³⁻⁵. Protocols such as the Rapid Ultrasound for Shock and Hypotension (RUSH) exists to guide users in searching for causes of hypotension in patients in a systematic fashion. Similarly, we propose a standardized, source-focused ultrasound workflow for sepsis that could potentially improve POCUS utility in sepsis diagnosis and treatment. We present a clinical case incorporating the SUPER protocol, a structured POCUS approach designed to rapidly identify infectious sources during early sepsis resuscitation in EDs.
Case Presentation
A 62-year-old male with chronic kidney disease on maintenance hemodialysis via a right internal jugular catheter presented with a fever of 38.9 degrees Celsius and chills for seven days and progressive dyspnea for two days. On arrival, he was hypotensive (blood pressure was 90/50 mmHg), tachycardic to 130 beats per minute, and drowsy. Providers were concerned for sepsis, but the initial history and clinical evaluation did not reveal an obvious source of infection.
A bedside POCUS SUPER protocol (Sepsis Ultrasound Protocol to identify source Etiology Rapidly) was performed during early resuscitation. The full protocol will be described in the discussion section and Figure 1. In this patient’s case, focused cardiac ultrasound demonstrated a mobile echogenic mass attached to the tricuspid valve, suggestive of infective endocarditis. (Figure 2). Three sets of blood cultures were obtained, and antimicrobial therapy was promptly tailored toward catheter-related bloodstream infection. Subsequent investigations confirmed right-sided infective endocarditis, and early ultrasound-based source identification facilitated timely, targeted management.
Discussion
Undifferentiated sepsis frequently prompts empiric, broad-spectrum antibiotic therapy while clinicians await definitive imaging or microbiologic confirmation. Evidence indicates that multi-organ POCUS improves diagnostic accuracy and shortens time to source identification, often within the first 10 minutes of ED assessment³⁻⁶. However, unstructured scanning may be inefficient in time-sensitive settings. The SUPER protocol was designed to provide a systematic, rapid, and repeatable POCUS framework focused specifically on identifying infectious sources in patients with sepsis rather than resuscitation physiology alone.
The SUPER protocol evaluates five key anatomical domains, selected based on the most common ultrasound-detectable causes of sepsis: (Figure 1)
- Cardiac – Assessment for infective endocarditis, particularly in patients with indwelling catheters or prosthetic valves
- Respiratory – Lung ultrasound for pneumonia, consolidation, and exudative pleural effusion
- Hepatobiliary/Gastrointestinal – Evaluation for cholecystitis, cholangitis, intra-abdominal collections, or perforation
- Genitourinary – Detection of hydronephrosis, pyelonephritis, or renal abscess
- Musculoskeletal/Soft Tissue – Identification of abscesses, septic arthritis, cellulitis, or necrotizing infection
Findings are documented in real time and integrated with clinical assessment and laboratory data. A prospective observational study in the ED is ongoing in our centre enrolling adults with clinically suspected sepsis in whom no source was identified after initial evaluation. Following stabilization, a structured SUPER protocol is performed at the bedside and completed within approximately 10 minutes, without interrupting ongoing resuscitation. Interim analysis of the ongoing study with a sample size of 70 patients suggests that the SUPER protocol demonstrates moderate sensitivity and specificity of 72.5% and 68.4 % respectively in identifying the sepsis source identification.
Evidence supporting POCUS in sepsis source identification continues to grow. Cortellaro et al. demonstrated that a POCUS-implemented approach identified the infectious source with approximately 73% sensitivity, outperforming initial clinical evaluation and significantly reducing time to diagnosis³. Subsequent reviews consistently show that lung, soft tissue, abdominal, and cardiac ultrasound provide the highest diagnostic yield in septic patients, particularly when conventional imaging is delayed or impractical⁴⁻⁶. Beyond source localization, POCUS contributes to sepsis care by guiding hemodynamic resuscitation, identifying sepsis mimics such as cardiogenic shock or pulmonary embolism, facilitating bedside source control procedures (e.g., abscess drainage and thoracentesis), and enabling serial reassessment to monitor treatment response, reinforcing the value of an integrated, multisystem ultrasound approach⁵⁻⁷. Although direct stewardship outcome data is limited, POCUS functions as a diagnostic accelerator that aligns closely with antimicrobial stewardship principles and represents an important area for future research studies⁸.
Conclusion
The SUPER protocol offers a structured, rapid POCUS approach for early source identification in undifferentiated sepsis. By integrating the protocol into ED resuscitation, it can enhance diagnostic confidence, support timely targeted therapy, and has the potential to advance antimicrobial stewardship. Further research is needed to define the SUPER protocol’s diagnostic performance and downstream impact on antibiotic use and patient outcomes.
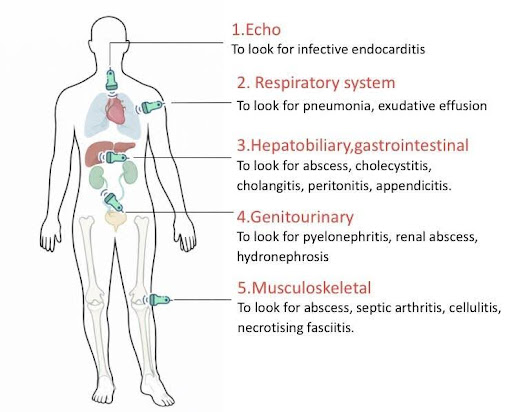
Figure 1: Five key anatomical domains of SUPER protocol based on the most common ultrasound detectable causes of sepsis.
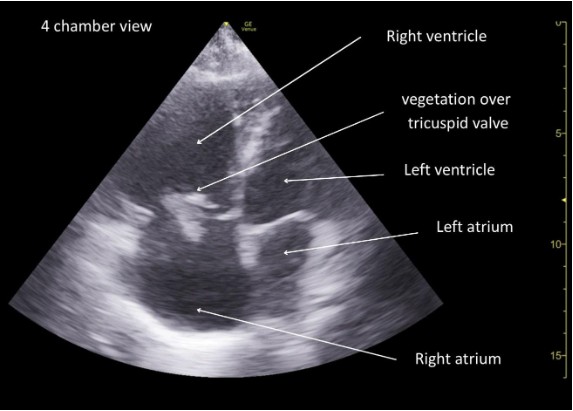
Figure 2: Focused cardiac ultrasound (4 chamber view) demonstrating a mobile echogenic mass attached to tricuspid valve suggestive of infective endocarditis.
References
- Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-810.
- Seymour CW, Gesten F, Prescott HC, et al. Time to treatment and mortality during mandated emergency care for sepsis. N Engl J Med. 2017;376(23):2235-2244.
- Cortellaro F, Ferrari L, Molteni F, et al. Accuracy of point-of-care ultrasound to identify the source of infection in septic patients. Intern Emerg Med. 2017;12(3):371-378.
- Verras C, Ventoulis I, Bezati S, et al. Point-of-care ultrasonography for the septic patient in the emergency department. J Clin Med. 2023;12(3):1105.
- Sweeney DA, Wiley BM. Integrated multiorgan bedside ultrasound for sepsis and septic shock. Semin Respir Crit Care Med. 2021;42(5):641-649.
- Alonso JV, Turpie J, Farhad I, Ruffino G. Protocols for point-of-care ultrasound in sepsis. Bull Emerg Trauma. 2019;7(1):67-71.
- Hobbs H, Millington S, Wiskar K. Multiorgan point-of-care ultrasound assessment in critically ill adults. J Intensive Care Med. 2024;39(3):187-195.
- Barlam TF, Cosgrove SE, Abbo LM, et al. Implementing an antibiotic stewardship program. Clin Infect Dis. 2016;62(10):e51-e77.
POCUS Clinical Images
Saddle Pulmonary Embolism Detected via Bedside Ultrasound
By Priyanka Pradhan, MD, David Murray, MD, FPD-AEMUS
Image Credit: Jenn Lee, MD
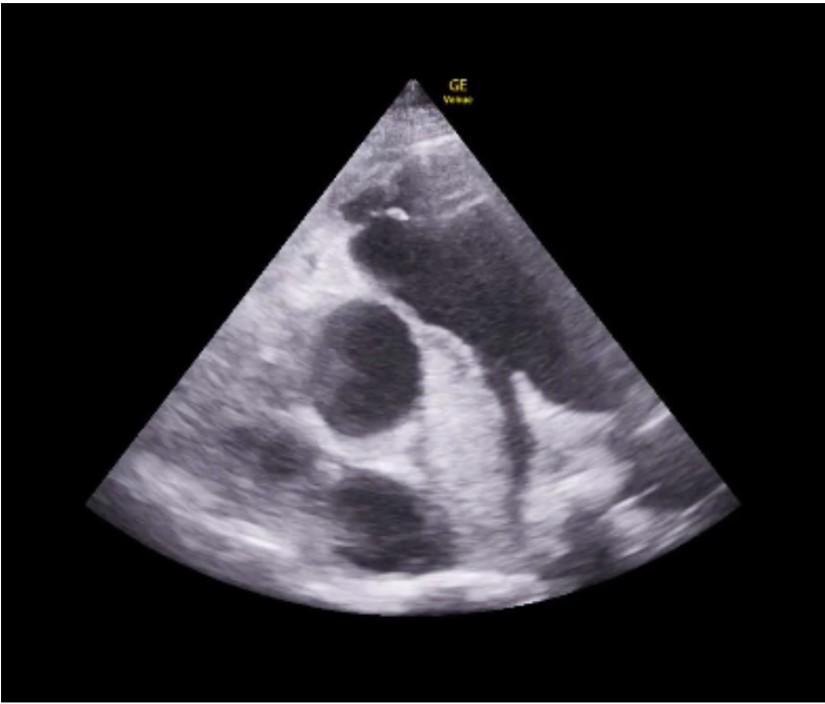
Figure 1
An 82 year old female presented to the Emergency Department (ED) for 1 week of left leg swelling and shortness of breath. In the ED, she was hypoxic to 88% requiring 30 liters per minute high flow nasal cannula and hypotensive to 69/44 mmHg requiring 5mcg/min norepinephrine drip. On exam, she was tachycardic to 127 beats per minute and tachypneic to 24 breaths per minute with labored respirations. Her lungs were clear to auscultation and she had bilateral 2+ leg edema with the left mildly greater than the right. Point-of-care transthoracic echocardiography showed a large non-mobile hyperechoic thrombus extending from the main pulmonary artery (PA) bifurcation and into the right main PA with right ventricular (RV) dilation. Computed tomography with contrast of the chest showed a large hypodense nonocclusive filling defect extending from the main PA into the right main PA which indicated acute pulmonary embolus (PE) with evidence of right heart strain (RHS) – the same diagnosis made within minutes by bedside echocardiography.
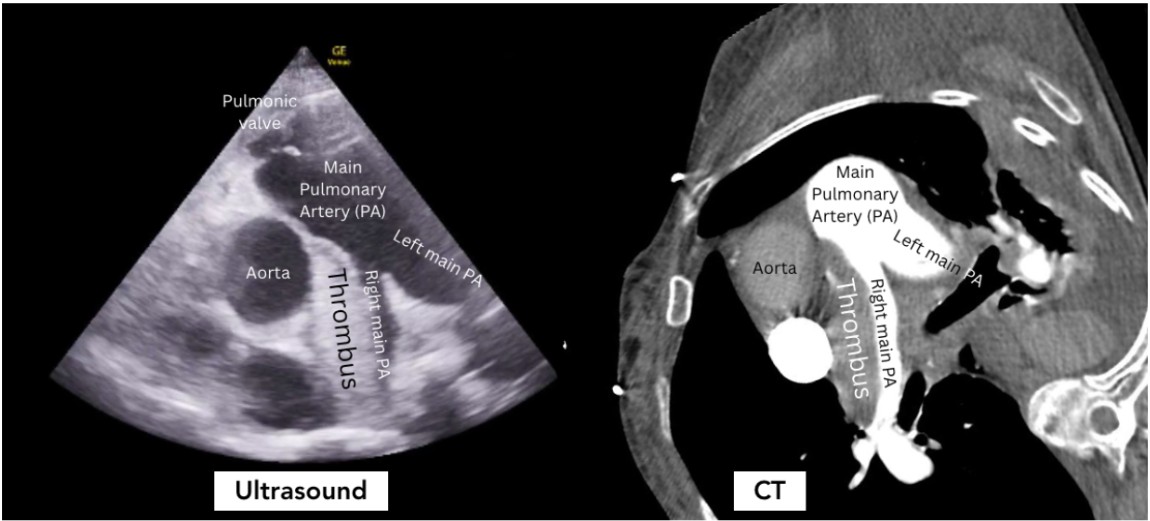
Figure 2
This case highlights the value of bedside echocardiography in the definitive diagnosis of a massive pulmonary embolism. Traditional point-of-care transthoracic echocardiography evaluates for indirect signs of acute PE through the identification of RHS 1,2. Generally, this is an assessment in parasternal long and apical views for RV dilation, McConnell’s sign (right ventricular free wall akinesis with sparing of the apex), and/or abnormal Tricuspid Annular Plane of Systolic Excursion (TAPSE <17mm is abnormal)2. The parasternal short view can show septal flattening (“D sign”) and RV dilation. More recent publications have demonstrated the utility of diagnosing RHS through the identification of a systolic notching pattern or abnormal pulmonary artery acceleration time in a right ventricular outflow track (RVOT) view3. Our case illustrates the value of the RVOT view for direct visualization of a large pulmonary embolism, allowing the emergency physician to definitively make the diagnosis of massive pulmonary embolism at the bedside, without the patient ever having to leave the resuscitation bay. This view is obtained by placing the probe in the parasternal short axis view and fanning cephalad until the right ventricular outflow tract is visualized.
References
- Dresden S, Mitchell P, Rahimi L, Leo M, Rubin-Smith J, Bibi S, White L, Langlois B, Sullivan A, Carmody K. Right ventricular dilatation on bedside echocardiography performed by emergency physicians aids in the diagnosis of pulmonary embolism. Ann Emerg Med. 2014 Jan;63(1):16-24. doi: 10.1016/j.annemergmed.2013.08.016. Epub 2013 Sep 27. PMID: 24075286.
- Alerhand S, Adrian RJ. What echocardiographic findings differentiate acute pulmonary embolism and chronic pulmonary hypertension? Am J Emerg Med. 2023 Oct;72:72-84. doi: 10.1016/j.ajem.2023.07.011. Epub 2023 Jul 10. PMID: 37499553.
- Afonso L, Sood A, Akintoye E, Gorcsan J 3rd, Rehman MU, Kumar K, Javed A, Kottam A, Cardozo S, Singh M, Palla M, Ando T, Adegbala O, Shokr M, Briasoulis A. A Doppler Echocardiographic Pulmonary Flow Marker of Massive or Submassive Acute Pulmonary Embolus. J Am Soc Echocardiogr. 2019 Jul;32(7):799-806. doi: 10.1016/j.echo.2019.03.004. Epub 2019 May 2. PMID: 31056367
Probe to Prostate: A POCUS Diagnosis of Prostatic Abscess
By Gillian Villasana, MD and Katheryn Larimore, MD
Case Report
Our patient is a 42-year-old male with a medical history of insulin-dependent diabetes (IDDM), hyperlipidemia, and end-stage renal disease (ESRD) on dialysis who makes urine two times per week, who initially presented to the emergency department after having purulent penile discharge that started about twelve days prior, now with suprapubic abdominal pain and fullness for the past several days. He was afebrile and vital signs showed heart rate of 97 beats per minute, blood pressure of 79/57 mmHg, respiratory rate of 18 breaths per minute, and oxygenation of 97% on room air. On exam, the patient had suprapubic abdominal tenderness without peritoneal signs. Patient deferred rectal exam. On laboratory testing, the patient had a white blood cell count of 20.5K. Urinalysis showed greater than 100 white blood cells, 84 red blood cells, many bacteria, and was negative for nitrates. Due to concern for infection and possible sepsis, intravenous vancomycin and cefepime were initiated in the ED. Straight catheterization was initially attempted without any return of urine. Point of care bladder ultrasound was done to evaluate bladder volume for urinary retention. It revealed a hyperechoic mass extending into the bladder consistent with a large prostatic abscess (PA), though bladder volume was approximately 68 mL, and foley catheter was later placed. (Figure 1) A computed tomography (CT) scan of the abdomen and pelvis with intravenous contrast demonstrated multiple rim-enhancing fluid collections which measured collectively as 5.3 x 5 x 5.6 cm with fat stranding over the prostate. Urology was consulted, and the patient was admitted to the hospital. He underwent a transurethral unroofing of the PA with urology the next day, and urine and blood cultures grew Escherichia coli.
Discussion
Prostatic abscesses are relatively rare. In the 1990s they were estimated as affecting 0.2-0.5% of males with a peak incidence of men aged 50s-70s years old1. The main risk factor for PAs is being immunocompromised, including by HIV, chronic renal failure, liver disease, chemotherapy use, immunosuppressive medications for organ transplants or rheumatologic conditions, and diabetes. Per Ackerman et. al, over 50% of patients with PAs have diabetes. Emergency physicians should be aware of PAs and recognize that they can be the initial presentation of a newly diagnosed chronic medical condition. Also, PAs can be complications of acute and chronic prostatitis, and prostatitis risk factors such as severe benign prostatic hypertrophy (BPH), neurogenic bladder, and chronic indwelling foley catheters or intermittent catheterizations can lead to PAs1,3. In fact, up to 5-10% of patients with acute prostatitis will develop PA if prostatitis is not adequately treated4. Thus, physicians should have heightened suspicion for PA in patients with prolonged symptoms or that are not responding to antibiotics3. In older men, a common etiology is having a recent prostate biopsy (8-11% of patients with PAs). Other risk factors include urethritis, epididymitis, anal receptive intercourse, and phimosis2. Prior to antibiotic use, the most common cause of PA was Neisseria gonorrhea in healthy men. However, since antibiotics have become ubiquitous, the most common bacteria (60-80%) are gram-negative, such as Escherichia coli1. The most common bacteria spread hematogenously is Staphylococcus aureus.
Patients often have symptoms of urinary tract infection, including dysuria and frequency, but can also present with signs of urinary obstruction, such as low back pain, perineal or suprapubic pain, tenesmus, or the feeling of incomplete voiding1. Sometimes patients may have only vague symptoms like generalized fatigue, fever, or sepsis. Up to one third of patients with prostate abscesses have urinary retention. Though our patient had the feeling of incomplete bladder emptying with suprapubic pain, his bladder was decompressed.
For diagnosis, more than 90% of patients with PAs exhibit leukocytosis, and nearly all will have leukocytes detected in the urine. On imaging, an abscess can be seen on transrectal ultrasound (TRUS), CT, or magnetic resonance imaging (MRI), with TRUS typically first line for diagnostic imaging1. Nevertheless, CT is more readily accessible in the ED and allows for better evaluation of fluid collections and gas, whereas CT and MRI better reveal spreading of the abscess into nearby tissues1,4. Furthermore, CT and MRI are more helpful for surgical planning due to the ability to make precise measurements and are more widely used in the United States4.
In our patient’s case, the use of point of care transabdominal ultrasound to investigate the patient’s sensation of urinary retention led to the preliminary diagnosis of PA. On ultrasound, in both the transverse (1) and longitudinal (2) view, a multiloculated abscess is seen, indicated by the hypoechoic areas, measuring approximately 5 x 5 x 5 cm. CT scan was then done to confirm and better characterize the abscess, urology was consulted, and ultimately the patient had definitive treatment with antibiotics and transurethral unroofing of the abscess in the operating room.
Conclusion
Though PA is a rare diagnosis, in an immunocompromised patient with symptoms of urinary retention and purulent discharge, bedside ultrasound may be used to evaluate the cause of the sensation of obstruction, which may reveal a PA. Our patient had several risk factors for PA, as well as known presenting symptoms and laboratory abnormalities, and a CT scan confirmed the abscess. Though bedside ultrasound may not be the most sensitive modality for diagnosing PA, it can be an effective use of resources and emergency physicians’ skills to expedite a patient’s diagnosis and treatment. With expedited diagnosis, the patient had the appropriate surgery, and antibiotics were then narrowed based the patient’s intraoperative fluid culture, with good overall outcome.
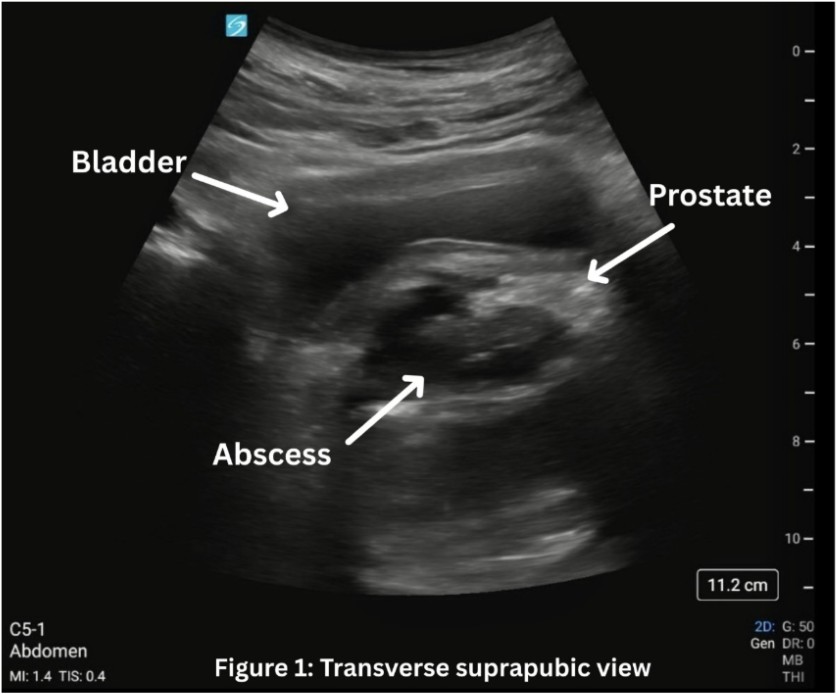
Figure 1: Transverse view of the bladder with prostate abscess.
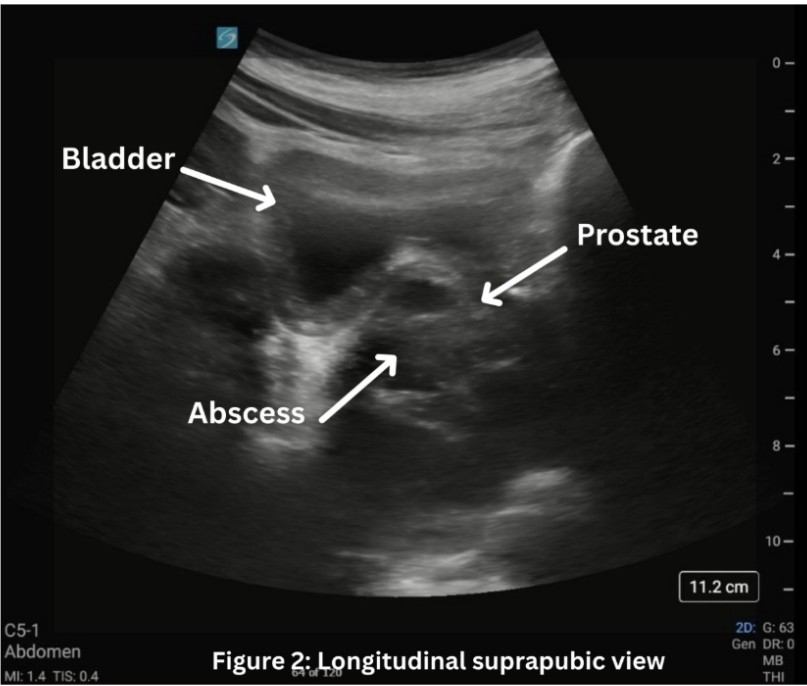
Figure 2: Longitudinal view of the bladder with prostate abscess.
References
- Ackerman AL, Parameshwar PS, Anger JT. Diagnosis and treatment of patients with prostatic abscess in the post-antibiotic era. Int J Urol. 2018;25(2):103-110. doi:10.1111/iju.13451
- Davis JE. Male Genital Problems. In: Tintinalli JE, Ma O, Yealy DM, Meckler GD, Stapczynski J, Cline DM, Thomas SH. eds. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 9e. McGraw-Hill Education; 2020. Accessed January 04, 2026. https://accessmedicine.mhmedical.com/content.aspx?bookid=2353§ionid=219643185
- Mercader D, Theophanous RG. Prostate abscess causing obstruction in an emergency department patient with constipation. JEM Reports. 2024;3(2):100084-100084. doi:https://doi.org/10.1016/j.jemrpt.2024.100084
- Wooster ME, Huang G, Sanders JW, Peacock JE. 1448. Prostate Abscess: Clinical Features, Management, and Outcomes of a “Stealth” Infection: A Case Series. Open Forum Infectious Diseases. 2019;6(Supplement_2):S528-S529. doi:https://doi.org/10.1093/ofid/ofz360.1312
Quadriceps tendon rupture in a patient with left knee pain
By Andrew G. Theophanous (MD Candidate) and Rebecca G. Theophanous, MD MHSc
University of Toledo College of Medicine; Duke University School of Medicine, Department of Emergency Medicine
Case Report
A healthy 34-year-old male presented with acute left knee pain and swelling after falling while playing basketball three days prior. The patient reported landing on his left knee and feeling a pop with immediate pain above the knee with subsequent swelling. He had a blood pressure of 155/85 mmHg, heart rate of 84 beats per minute, and was afebrile. On examination he had severe tenderness at his left distal thigh with a deformity superior to his patella. He had intact strength, sensation, and distal pulses in both legs but could not passively extend his left distal leg. Point-of-care ultrasound (POCUS) demonstrated a disrupted left distal quadriceps tendon with anechoic fluid, tendon enlargement near the insertion site, and tendon fiber distortion consistent with a complete tendon rupture. Left knee x-ray showed a joint effusion with soft tissue swelling anterior to the patella but no acute fracture.
The patient was discharged in an extension knee brace, with outpatient magnetic resonance imaging (MRI) confirming a complete left quadriceps tendon rupture with surrounding fluid, hematoma, and tendon retraction. He underwent urgent orthopaedic surgery with a good functional outcome.
Discussion
Quadriceps tendon rupture can occur in emergency department (ED) patients with trauma or in patients with underlying pathology.[1,2,3] It is more common in patients over age 40 years old or with comorbidities including rheumatoid arthritis, systemic lupus erythematosus, gout, chronic kidney disease, secondary hyperparathyroidism, peripheral vascular disease, and diabetes mellitus.[4] Predisposing risk factors include degenerative tendon changes, tendon overuse injuries, previous injury, and partial tendon tears.[1,3] Complete quadriceps tendon rupture is diagnosed clinically by distal thigh tenderness, a suprapatellar hematoma, and inability to extend the distal leg against gravity.[1,2] MRI is the gold standard for diagnosis but is difficult to obtain in the ED or clinic.[3,4] Thus, tendon rupture diagnosis can be augmented by POCUS. POCUS findings include distortion of tendon fibers near the patellar insertion site, tendon enlargement, surrounding anechoic fluid, and tendon retraction (Figures 1 and 2).[3,4] Physicians should recognize clinical signs of tendon rupture and understand how to incorporate POCUS to expedite diagnosis and guide appropriate urgent surgical management.[4,5] Urgent orthopaedics surgical repair for complete ruptures is key to prevent permanent damage and disability, thus patients require close orthopaedics follow up.[4,5]
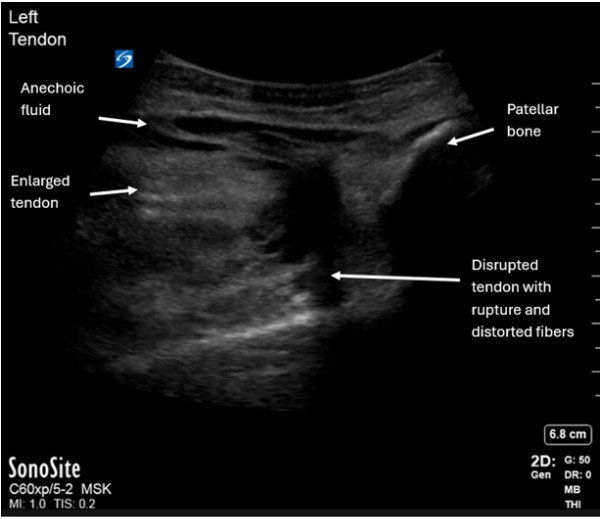
Figure 1: Longitudinal point-of-care ultrasound with complete rupture and retraction of left quadriceps tendon (white arrows) away from the patellar bone.
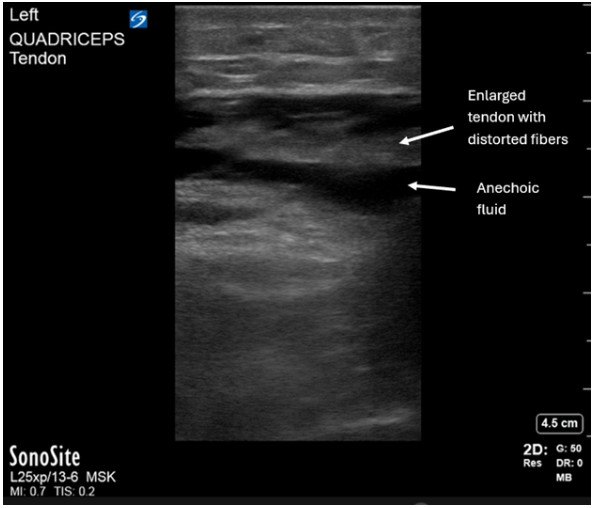
Figure 2: Point-of-care ultrasound longitudinal view of an enlarged hypoechoic quadriceps tendon with distortion of normal tendon fibers and surrounding anechoic fluid.
References
- InformedHealth.org [Internet]. Cologne, Germany: Institute for Quality and Efficiency in Health Care (IQWiG); 2006-. Overview: Tendon overuse injuries (tendinopathy) [Updated 2022 Mar 29]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK555501/.
- White AE, Uppstrom TJ, Pascual-Leone N, et al. Quadriceps Tendon Turndown for the Treatment of Chronic Quadriceps Tendon Insufficiency: Surgical Technique. Arthroscopy. Techniques. Technical Note: Knee. 2025 April;14(4): 103338. https://www.arthroscopytechniques.org/article/S2212-6287(24)00498-5/fulltext
- Quzli AA, Ali-Qazalbash ZM, Alkhatib SA, et al. Quadriceps Tendon Ruptures: A Clinical Review. Cureus. 2025 Oct 30;17(10):e95757. doi: 10.7759/cureus.95757. https://pubmed.ncbi.nlm.nih.gov/41181796/
- Arnold EP, Sedgewick JA, Wortman RJ, et al. Acute Quadriceps Tendon Rupture: Presentation, Diagnosis, and Management. JBJS Rev. 2022 Feb 7;10(2). doi: 10.2106/JBJS.RVW.21.00171. https://pubmed.ncbi.nlm.nih.gov/35130193/
- Touhey DC, et al. Chronic Quadriceps Tendon Rupture-Surgical Treatment and Outcomes: A Systematic Review. J Knee Surg. Thieme Medical Publishers Inc.. 9 May 2025. www.thieme-connect.com/products/ejournals/abstract/10.1055/a-2585-5037.
POCUS in Global Health
Lights in Malawi (medical student perspective)
By by Hannah Lynch, MS2 at University of Arizona College of Medicine Phoenix
Our first sunset in Malawi was magnificent. If you’ve never seen the sun fall behind the legendary “Mountains Beyond Mountains” that inspired Paul Farmer, I implore you to use your imagination, although the image you have conceived still does not compare to the real thing. The hazy evening light accompanied by the occasional buzzing, potentially infectious mosquito, tickles the soft hair on your arms and sends shivers up your spine. The sky faints away after a long, brilliant day into a sea of maroon, then the pitch-black falls and the air chills.
Earlier that day, 600 women and children stood like dominoes in a line that wound from the steps of the church to the back road where we had parked our bus, toe to heel, waiting on blood tests, physician visits, and medication dispersals. We had shown up anticipating 200-300 orphans and caretakers from our partnering childcare institutions and were entirely impressed and overwhelmed with the sheer number of patients who streamed in from nearby villages. Our four physicians from the United States and four Malawian Healthcare Workers fervently worked to see every patient with the dedication of a small army. Meanwhile, the pharmacy students were locked away in our makeshift medical room, scrambling to locate medications and scribbling hieroglyphic prescriptions in faded ink on the edge of paper bags for parents and caregivers. Children and young women gathered near the dilapidated building-turned-clinic until we could no longer see the steps leading up to the door. They sat and watched with sleepy eyes, slowly making a path for their friends and neighbors to retrieve medications. It was barely late afternoon when the word began to spread, “Get in the bus! We need to make it back before nightfall.”
Though we still had 200+ pharmacy prescriptions to fill, our driver and local associates urged us to stop what we were doing and pack up our things. “We can bring back the medications and pass them out tomorrow. Get in the bus. Now.” And so, unfamiliar with the territory, we hustled into the rusting, well-loved school bus with an uneasy urgency and spoke in hushed tones as we watched the sky begin to turn from its bright airy brilliance to every hue of pink and gold. My eyes were glued to the windows. I fought the post-clinic fatigue and held my breath as the last ray of sun dipped below the highest mountain, leaving the geography to its dusky soft glow. “Mountains Beyond Mountains” echoed between my ears.
Then, the dark fell. It really did fall, too. An unexpectedly abrupt decline of the light allowed us only to see a few feet in front of us with the lights of the bus. Retinal rods were highly undervalued in my life before this moment. I’d never seen such widespread darkness. No wonder we were in such a hurry to get home.
It did not occur to me until we re-entered the town that the light I had grown so accustomed to throughout my whole life was not normal. In truth, this rural town was a more accurate version of “real life”, without the luxury of electricity to light my way, spaced out appropriately by city planners before I was born. Instead, life continued in the pitch black, dotted with the occasional flare of wealthy fluorescence or cooking fire. A flash of a pink bow was all that revealed a child as she dipped dangerously close to our bus, and the motion of a friend’s wave to our driver gave away the bustling life around us. How do they live like this, I wondered as I fished out my phone to type away the marvel of what I had seen, squinting at the LED screen in poetic irony.
The next day, I found myself slightly less busy, with only 400 patients to see. I found one of the attendings and asked if there was anything I could do to help. “I brought a portable ultrasound machine and have some gel in my bag. I’d love to find an opportunity to use it.” She quirked an eyebrow at me.
“Is anyone trained? I’ve never used one before.”
“Actually, yes! I am.” I proudly held the probe up to her inspection and grinned when she called me over to scan a patient’s abdomen. She stared at the phone screen from behind me as I manipulated the device, pointing out the differences between fluid and adipose tissue. “How do you know what you’re looking at?”
“The sound waves bounce off liquids and solids and return to the probe. They all look different…” I explained as much as I could while I recorded the image. “You can see some really cool things with this! It’s not invasive, doesn’t require any medications, and it’s portable, so you can bring it anywhere.”
She nodded in agreement and swiftly sent our patient off to the hospital for a non-emergent surgical procedure to fix the problem, based on her clinical judgement and my ultrasound images. “We’ve never had one of those to use out here. I suppose we’re still in the dark ages of medicine. In the U.S., we practice medicine under harsh fluorescent lighting. In Malawi, it feels like we practice medicine in the dark. It’d be nice to be able to see a little more clearly, sometimes.”
I thought of all the patients who wouldn’t need to commute 12 hours to the nearest hospital, parents who would rest knowing that their child’s illness would self-resolve, and children who would make it to adulthood because their time-sensitive emergency would be discovered. The cost of the Butterfly ultrasound probe I was using (grandiose as it is for a medical student) felt small in comparison.
I am learning to become a physician during a time when the U.S. coordinates its exit from the grand stage of humanitarian aid. Our scientific advances have become so advanced that we have forgotten what it’s like to live in a time when the sun went down and there were no fluorescent lights, miracle cures, or lifelong disease protection. My personal beacon of light throughout the dark days of practicing medicine in a country that undervalues the importance of public health has become the tiny-ultrasonic-beam-machine that I can fit in my pocket, connect to my phone, and use to circumvent some of the costs of chasing down a diagnosis (when clinically indicated).
The light in the eyes of a patient when we offer to perform an ultrasound scan at this visit, instead of sending them to an imaging facility and making another appointment, is contagious. These are the moments that make my medical school debt feel worth it.
Ultrasound is not the solution to our cumulative global health inequalities. The physicians in resource-poor countries like Malawi have extraordinary abilities to use their clinical experience to make robust differential diagnoses without the use of imaging tools. I am always so impressed by them. Simultaneously, I am growing increasingly excited for the motivated medical students of my generation, who are being trained and equipped with point-of-care ultrasound machines, to go out into the places where the night is painted with stars instead of streetlamps and do our best to make a difference.
Advancing Point-of-Care Ultrasound Education in Rwanda: Lessons Learned, Challenges, and the Path Forward
By Timothy J. Batchelor, MD MBA; Patrick Lanter, MD MPH; Enoch Obeng, MD; Eric NSENGIYUMVA, MD; GIHANA Charles Wilson, MD; Sarah Shenge, MD; Pascal MUGEMANGANGO, MD; Doris Lorette UWAMAHORO, MD; and Michelle Feltes, MD MACM
Point-of-care ultrasound (POCUS) has become increasingly central to high-quality emergency care and its integration continues to reshape emergency medicine (EM) worldwide. POCUS offers an essential diagnostic lifeline in resource-limited environments where conventional radiographic imaging is difficult to obtain or unavailable – but its true impact depends on comprehensive training and ongoing quality assurance. In Rwanda, POCUS implementation has advanced significantly thanks to a dedicated partnership between the University Teaching Hospital of Kigali (CHUK), the University of Rwanda, and Stanford Emergency Medicine’s International and Ultrasound sections. This joint effort has fostered the development of a comprehensive POCUS training and quality assurance program embedded within the EM residency at CHUK.
Prior to this initiative, while Rwandan EM residents used ultrasound in clinical care, there was minimal formal structure for POCUS education or image quality review. With initial seed funding acquired in 2022, the program launched with the acquisition of portable ultrasound probes and the creation of a two-part bootcamp-style curriculum. This in-person curriculum was complemented by a robust remote quality assurance system leveraging Butterfly Enterprise software, and a virtual longitudinal program designed to promote continued skills acquisition. Resident scans are reviewed by Stanford Ultrasound Faculty or Fellows and feedback is delivered rapidly by email and mobile alerts, enabling ongoing skill improvement. These efforts have not only improved competency at the bedside but have also inspired a culture of clinical and scholarly curiosity among trainees.
A cornerstone of the program’s sustainability has been the cultivation of local “superusers” – Rwandan residents who have demonstrated advanced skills and leadership in POCUS. The superuser pathway includes intensive biweekly virtual QA sessions, opportunities to develop and teach new didactic content, and a leading role in hands-on mentorship. While the original vision was for these superusers to take the helm of the program at CHUK after graduation, the realities of the Rwandan health system have posed a formidable challenge. The first cohort of superusers have since graduated but dispersed to other institutions within the country, leaving a gap in on-site leadership continuity. As a result, ongoing involvement from Stanford’s remote faculty and recurring in-person training cycles remain vital to the health of the program.
Additional challenges have further highlighted the barriers of global health innovation. Equipment reliability and maintenance have emerged as significant hurdles. With no local product support from the ultrasound manufacturer, broken equipment must be transported back to the United States for repair and then re-deployed to Rwanda – a process that is both time-consuming and logistically daunting. Internet connectivity is another persistent obstacle: while local data plans suffice for batch image uploads, insufficient bandwidth precludes real-time teaching via teleguidance, a feature that remains aspirational. The logistical complexity of continually reloading data plans, often in person by the Rwandan superusers, adds another layer of burden to maintaining clinical workflow.
Despite these obstacles, steady progress continues. Each new residency class participates in refined boot camps and undergoes skills and knowledge assessments designed to measure POCUS impact on real-time clinical decision-making. Streamlined documentation workflows and increased administrative support from CHUK leadership have driven greater integration of ultrasound into resident training and daily clinical practice. Creative problem solving by residents and faculty, including technical training enhancements and the establishment of rapid-response support via WhatsApp by Stanford partners, has allowed the team to respond nimbly as challenges arise.
Recently, the program achieved a significant milestone, receiving additional grant funding to support phase two of the remote POCUS education project, expanding the research and sustainability components of this effort. Funds will be allocated to evaluate how POCUS use influences diagnostic reasoning and patient management in the ED. By collecting and analyzing data on how ultrasound changes clinical decisions and patient outcomes, the critical evidence base needed for continued program expansion can be obtained. This next phase will not only strengthen training at CHUK but also inform advocacy efforts to bring sustainable POCUS capacity to a wider network of healthcare institutions throughout East Africa.
The integration of POCUS in Rwanda is both a story of ongoing success and evolving complexity. While barriers remain – from equipment logistics to workforce retention – the collective commitment of Rwandan and international partners is unflagging. With each training cycle and research milestone, the vision of local, sustainable, and high-quality POCUS for emergency care in Rwanda comes into sharper focus.